
In the field of biotechnology, understanding "How Does a Peptide Synthesizer Work" is crucial. Dr. Emily Thompson, a leading expert in peptide chemistry, emphasizes, "Precision in peptide synthesis is vital for effective drug development." This underscores the importance of the synthesizer's role in accuracy and efficiency.
Peptide synthesizers automate the production of peptides, essential for research and therapeutic applications. They utilize solid-phase synthesis, a method invented in the 1960s. The process involves the stepwise addition of amino acids while monitoring purity and yield. Each cycle requires careful calibration, influencing the final product's quality.
However, not all synthesizers guarantee optimal results. Factors like temperature, reagents, and synthesis time can impact outputs. These elements remind us of the complexities in peptide synthesis. Despite advancements, there are challenges that require continuous improvement and knowledge. Understanding these intricacies can enhance both research and healthcare methodologies.
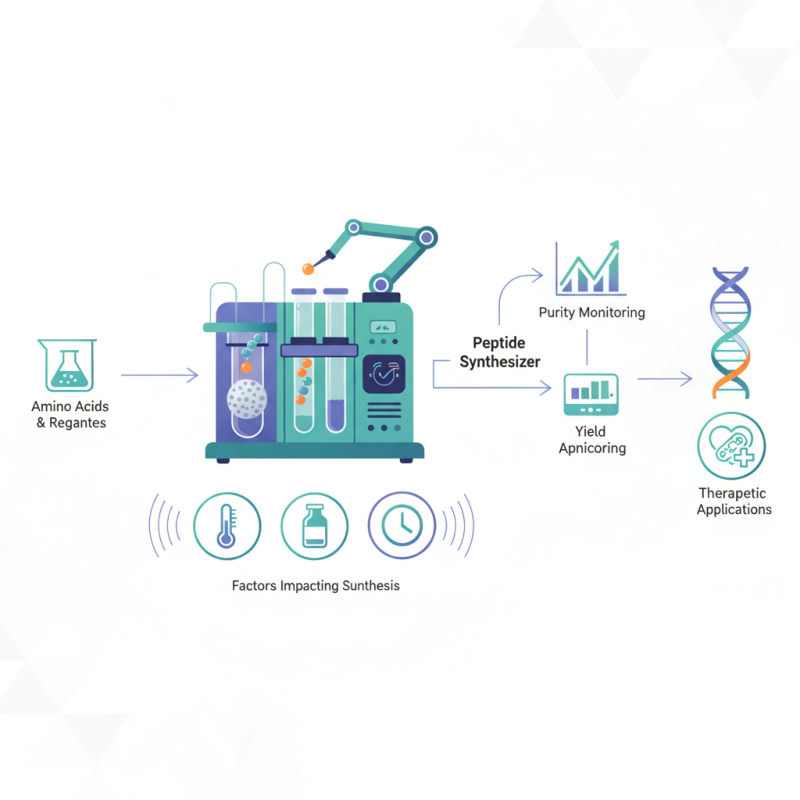
Peptide synthesis is a complex but intriguing process. It starts with the assembly of amino acids, building blocks of peptides. A peptide synthesizer automates this assembly. This machine uses a solid-phase technique to attach amino acids in a controlled sequence. A resin supports these amino acids, facilitating their chain formation.
Tips: Ensure the purity of amino acids. Impurities can derail synthesis. Regularly calibrate your synthesizer for accuracy. Small fluctuations can lead to significant errors in peptide chains.
The process involves several steps: activation, coupling, and deprotection. Activation prepares amino acids for linkage. Coupling joins them together, while deprotection removes protective groups to enable further reactions. Each step requires precision. Any miscalculation might affect the final product’s functionality.
Tips: Document each step meticulously. A clear record will help identify issues if something goes wrong. Regular troubleshooting can guide you back on track. Patience is vital; peptide synthesis is an iterative process that often requires adjustments and reevaluation for success.
Peptide synthesizers are vital tools in biochemistry. Understanding their key components can enhance your research outcomes. A solid understanding of these components ensures reliable results and enhances the efficiency of peptide synthesis.
The primary component of a peptide synthesizer is the reaction chamber. This area allows for the sequential addition of amino acids. Accurate temperature control is crucial here. It impacts the coupling efficiency. Another essential part is the solvent delivery system, which manages solvent and reagent flow. Regular maintenance is necessary to avoid blockages and ensure consistent operation.
Monitoring systems play a significant role as well. They track the reaction progress in real-time. This feedback can help identify if the synthesis is progressing as expected. In some cases, inconsistencies may arise. Issues such as incomplete coupling can occur due to suboptimal conditions. Addressing these irregularities is critical for improving the quality of your peptides. Each component must work harmoniously for optimal performance.
Peptide synthesizers are crucial in producing high-quality peptides for research and therapeutic use. Optimizing the process can significantly enhance yield and purity. According to a report by the Protein Society, the efficiency of peptide synthesis can be improved by employing parallel synthesis techniques. This method allows simultaneous synthesis of multiple peptides, reducing time and resource expenditure.
One key optimization technique is the choice of coupling reagents. Selecting the right reagents can increase the reaction efficiency and reduce side products. A study published in Bioorganic & Medicinal Chemistry noted that using specialized coupling agents could raise yields by up to 30%. Another influential factor is the thorough purification of peptides post-synthesis, often utilizing reverse-phase chromatography. This step greatly enhances purity levels, making the peptides more suitable for further applications.
Tips: Regularly calibrate your synthesizer to ensure precise measurements. Always evaluate the purity of your initial reagents, as impurities can skew results. Reflect on your purification methods; even minor adjustments can yield significantly different outcomes in peptide quality.
| Optimization Technique | Description | Expected Yield (%) | Purity Level (%) |
|---|---|---|---|
| Temperature Control | Maintaining optimal temperature during synthesis to reduce side reactions. | 85 | 90 |
| Reaction Time | Optimizing reaction time to allow complete synthesis without degradation. | 80 | 92 |
| Reagent Quality | Using high-purity reagents to minimize impurities in the final product. | 90 | 95 |
| Coupling Cycle Optimization | Adjusting the number of coupling cycles based on peptide length. | 75 | 88 |
| Post-synthesis Purification | Implementing techniques like HPLC for better purity. | 88 | 97 |
Peptide synthesis can present various challenges. Monitoring these issues is crucial for effective results. A common problem is incomplete coupling reactions. This occurs when amino acids fail to bond properly. You might notice lower yields during your synthesis process. It's vital to check the coupling reaction times and temperatures. Adjust them based on your specific peptide sequence.
Another frequent issue is the formation of impurities. Impurities can arise through side reactions or failed synthesis steps. Regularly analyzing samples via mass spectrometry can help. This provides insights into the purity and identity of your peptides. Remember, even small variations can lead to significant changes in outcomes.
Additionally, consider the flow rates during synthesis. Too fast or too slow can hinder results. Be prepared to tweak these settings. You may find that minor adjustments lead to improved yields. Reflecting on previous runs can highlight areas for refinement. Accumulating data over time will enhance both your skills and your synthesizer’s performance.
Recent advancements in peptide synthesis technology have significantly transformed the field. With a growing demand for peptides in pharmaceuticals, the market is projected to reach over $30 billion by 2025, according to industry reports. This growth reflects an increasing reliance on peptide-based drugs, which offer high specificity and lower side effects compared to traditional therapies.
Innovations in solid-phase peptide synthesis (SPPS) and automated synthesizers enhance efficiency. New methodologies have emerged, allowing for the synthesis of complex peptides with improved yields. Despite the progress, challenges persist. The synthesis of larger and more intricate peptides remains labor-intensive. The need for enhanced purification methods is also critical to ensure product quality.
Emerging trends focus on integrating artificial intelligence with peptide synthesis. AI-driven algorithms can optimize synthesis parameters, potentially reducing errors. However, this approach requires further validation. Additionally, bioconjugation techniques are evolving, expanding applications in drug delivery systems. While the future holds promise, caution is needed. Continuous research and development are essential to fully realize these technologies’ potential and address existing limitations.
